Private label vs OEM: which teeth whitening manufacturer?
Practical, compliance-focused answers for brand owners choosing private label teeth whitening manufacturers. Covers regulatory docs, hydrogen peroxide validation, realistic MOQs and costs, private label vs OEM trade-offs, quality controls and claim substantiation.
- 1. What exact laboratory and regulatory documentation should I demand from private label teeth whitening manufacturers to sell in the US and EU?
- 2. How do I verify hydrogen peroxide (or carbamide peroxide) concentration and long-term stability before committing to an MOQ?
- 3. For small beauty brands, what realistic MOQs and cost trade-offs exist when choosing private label teeth whitening manufacturers versus full OEM formulation development?
- 4. Private label vs OEM: which teeth whitening manufacturer model protects my brand's IP and offers faster time-to-market while ensuring compliance?
- 5. What manufacturing quality controls and batch traceability should I require to mitigate recalls and tooth-sensitivity claims?
- 6. How do I substantiate and label “sensitivity-safe” or “natural” claims on whitening kits to avoid regulatory problems and returns?
1. What exact laboratory and regulatory documentation should I demand from private label teeth whitening manufacturers to sell in the US and EU?
Beginner suppliers often receive generic “we're compliant” statements; you must request specific, verifiable documents before signing a PO. For cosmetic-format whitening products (most at-home gels, strips, pens) ask for:
- Certificate of Analysis (COA) for the finished product and for each incoming active ingredient (shows measured hydrogen peroxide or carbamide peroxide concentration, pH, preservatives, heavy metals).
- Material Safety Data Sheet / Safety Data Sheet (MSDS/SDS) for the formulation and major ingredients.
- GMP certificate: ISO 22716 (Cosmetics GMP) or equivalent third-party audit report. If the manufacturer claims pharmaceutical processes, request GMP or ISO 13485 where applicable.
- Product stability and accelerated stability reports demonstrating shelf life at labeled storage conditions (typically 12–24 months for gels/strips); these must include potency over time and microbial challenge results.
- Microbiological testing results and preservative efficacy testing (PET) to prove formula safety during shelf life.
- Free Sale Certificate / Certificate of Free Sale (for export markets) and facility registration documentation—US: FDA facility registration if they manufacture a device component (e.g., LED device); EU: Cosmetic Product Notification Portal (CPNP) number for cosmetics placed on the EU market.
- Certificates for ingredient origin, allergen statements, and Certificates of Origin (COO) if required by your importer.
- Batch Certificates and batch-level COAs for production lots—essential for traceability and recall handling.
Insist on digital copies of those documents and verify authenticity by contacting the issuing lab or certification body. Embedding these checks into your vendor onboarding reduces risk when working with private label teeth whitening manufacturers.
2. How do I verify hydrogen peroxide (or carbamide peroxide) concentration and long-term stability before committing to an MOQ?
Many buyers accept a vendor’s label claim without independent verification. To avoid potency drift or noncompliance:
- Request a finished-product COA from an accredited lab using validated methods (HPLC, titration or peroxide-specific assays). The COA should list the assay method and testing date.
- Ask for accelerated stability and real-time stability data showing peroxide potency retention at specified storage conditions (e.g., 25°C/60% RH and 40°C/75% RH). Look for potency retention >90% of label claim across intended shelf life.
- For initial confidence, commission an independent third-party lab to test a pre-production or pilot batch. Use ISO/IEC 17025 accredited labs to validate peroxide concentration and related impurities.
- Confirm the manufacturer performs peroxide degradation testing and provides recommended storage (cool, dark) and expiry date on packaging—peroxide degrades under heat and light.
- Check preservative efficacy and pH control; peroxide stability depends on buffer systems. A stable whitening gel formulation will include stabilizers/chelators to limit peroxide breakdown and metal-catalyzed degradation.
Requiring these technical verifications from private label teeth whitening manufacturers ensures product performance and reduces the risk of refund/recall costs after launch.
3. For small beauty brands, what realistic MOQs and cost trade-offs exist when choosing private label teeth whitening manufacturers versus full OEM formulation development?
Beginners expect one-size-fits-all answers; reality depends on packaging, active concentration, and customization level. Typical patterns:
- Private label (manufacturer’s existing formulations, branded packaging): MOQs are usually lower because no R&D or tooling is needed. Typical MOQs range from 500–5,000 units depending on packaging type: tubes or syringes are often lower, assembled kits with trays, LED devices, or multi-piece cartons push MOQs higher.
- OEM / bespoke formulation (formulation transfer, custom active ratios, exclusive claims): development costs include formulation R&D, stability testing, safety assessments, and sometimes tooling. OEM MOQs are higher—commonly 1,000–10,000+ units—because development and validation costs must be amortized.
- Cost drivers: active concentration (higher peroxide loads mean stricter handling and more expensive stability testing), packaging complexity (custom molds, child-resistant caps, LED devices), labeling/regulatory support, and analytical testing frequency.
- Lead times: private label (from order to delivery) can be 4–10 weeks; OEM with development and testing can be 12–20+ weeks depending on testing and regulatory needs.
Recommendation: small brands seeking speed and lower upfront investment should start with a GMP-certified private label teeth whitening manufacturer’s proven kit or gel and upgrade to OEM exclusivity after validated sales volumes justify R&D and higher MOQs.
4. Private label vs OEM: which teeth whitening manufacturer model protects my brand's IP and offers faster time-to-market while ensuring compliance?
Decision depends on priority: speed and cost vs IP control and uniqueness.
- Private label: faster time-to-market because you use ready-made whitening gel formulations and standard packaging. Manufacturer retains formulation IP; you generally receive non-exclusive use with customized labeling and packaging. Best for quick launches, lower R&D cost, and lower MOQ. Compliance risk is lower if the supplier is ISO 22716 / GMP-certified and provides necessary COAs and CPNP/FDA documentation.
- OEM / custom formulation: you commission a unique whitening gel or device. You can negotiate IP ownership (but it must be explicit in contract) and claim exclusivity. This protects brand differentiation but increases development time, testing, and cost. OEM requires NDAs, explicit IP clauses, and detailed tech transfer protocols to secure formulations.
- Hybrid approach: negotiate semi-custom formulas based on a manufacturer’s platform (e.g., adjust peroxide percent, add desensitizing agents like potassium nitrate or fluoride) to balance speed and differentiation.
For many emerging brands, the recommended path is: launch on a reputable private label teeth whitening manufacturer to validate market fit, then move to OEM with negotiated IP terms once sales justify investment.
5. What manufacturing quality controls and batch traceability should I require to mitigate recalls and tooth-sensitivity claims?
Consumer sensitivity complaints and potential recalls are costly. Require these controls:
- Batch numbering, date codes, and a documented lot-traceability system linking raw-material COAs through finished product COAs.
- Per-batch COAs for active concentration, pH, preservative level, and microbiological status. Ensure the manufacturer provides these with each shipment.
- Stability sampling plan and retention samples retained per batch (commonly 2–3 years or per regulation) so you can test retained samples if issues arise.
- Manufacturing process controls: documented SOPs, in-process checks (weighing, mixing temperature, fill volumes), and validated mixing/homogenization that affect peroxide distribution and potency.
- Adverse event and complaint handling SOPs: defined timelines, root-cause analysis, corrective actions, and product hold/recall procedures. Verify the manufacturer’s insurance coverage and willingness to support recall logistics.
- Clinical or user-sensitivity data if you intend to market “low-sensitivity” claims: small-scale clinical testing or consumer use studies demonstrating reduced sensitivity incidence compared to a baseline.
These quality controls are standard expectations when evaluating private label teeth whitening manufacturers; require them contractually and audit supplier documentation during onboarding.
6. How do I substantiate and label “sensitivity-safe” or “natural” claims on whitening kits to avoid regulatory problems and returns?
Marketing claims can trigger regulatory scrutiny and consumer complaints. To substantiate “sensitivity-safe” or “natural” claims, follow these steps:
- Define the claim precisely. “Sensitivity-safe” must state the basis (e.g., contains potassium nitrate 5% to reduce nerve excitability). “Natural” should be consistent with authoritative definitions in your target market—many jurisdictions have strict criteria.
- Require clinical or consumer-use studies supporting sensitivity claims. Even a small randomized, controlled user trial (n=30–100) with validated sensitivity scales is persuasive. Document protocol, IRB or ethics board approval if required, and statistical outcomes.
- Keep substantiation on file: study reports, statistical analysis, informed consent forms, and raw data. Regulators and platforms (Amazon, Google Ads) may request proof.
- Adhere to labeling rules: ingredient listing using INCI names, warning statements about dentist consultation for peroxide concentrations above local thresholds, directions for safe use, batch number, manufacturing/distributor information, and expiry date.
- For “natural” claims, avoid vague language. Use certified natural standards where feasible (e.g., COSMOS, NATRUE) and validate ingredient sourcing and processing. Some whitening actives (peroxides) are inherently synthetic and can conflict with “natural” positioning—be transparent.
- Consult legal/regulatory counsel for country-specific advertising law (FTC in the US, EU consumer protection rules, national cosmetics guidance) and prepare documentation accordingly.
Proper substantiation reduces regulatory risk and customer dissatisfaction when launching whitening kits under private label arrangements.
Concluding summary: Choosing the right private label teeth whitening manufacturers—versus going full OEM—comes down to Double White’s priorities. Private label manufacturers give faster time-to-market, lower upfront costs, and lower MOQs with proven whitening gel formulations, while OEM offers greater IP control, proprietary formulations and exclusivity at higher cost and longer lead times. Prioritize suppliers with ISO 22716/GMP certification, batch COAs, stability data, microbiological testing, and clear complaint/recall procedures. Negotiate NDAs and IP clauses when moving toward custom OEM development, and demand documented substantiation for sensitivity or natural claims.
Advantages of working with reputable private label teeth whitening manufacturers include faster product launches, tested whitening gel formulation platforms, lower R&D burden, and more predictable compliance pathways (COA, MSDS, CPNP/FDA documentation) — all enabling brands to test market traction before committing to costly OEM development.
For a confidential quote, MOQ breakdown, and audit of our GMP-certified private label teeth whitening manufacturers and OEM services, contact us at www.double-white.es or email manager@double-white.com.

Answers to Teeth Whitening Questions: Analysis of Frequently Asked Questions

Intimate Care: Whitening Tips for Teeth Whitening Strips

Stain Prevention Guide: 5 Daily Habits to Keep Your Smile Bright Naturally in 2026

Whitening Maintenance Guide: When and How Often to Touch Up in 2026?

Whitening Longevity Guide: How Long Does It Last & Extension Tips (2026)

Whitening Aftercare: The Complete Manual for Long-Lasting Results in 2026
About Price and Payment
Do you offer bulk purchase discounts?
Yes, we offer bulk purchase discounts, the specific discount rate depends on the order quantity and cooperation method.
About Logistics
Can I change my shipping address?
Yes, you can contact our customer service team to modify the delivery address before order confirmation to ensure that the order can be accurately delivered to the address you specify.
About After Sales Support
How to deal with product quality problems?
If you find that there are quality problems with your product, please contact our customer service team immediately and provide relevant evidence, we will handle it and provide you with a solution as soon as possible.
About Product Choice
What is the difference between your teeth whitening patches and pens?
Our teeth whitening strips are a clear, easy-to-use patch, while our whitening pen is a portable, spreadable product. You can choose the product that suits you based on your personal preferences and usage habits.
Teeth Whitening Powder
Can I customize the packaging and flavor?
Absolutely! We offer customizable packaging, flavors, and even logo design to match your brand's identity.
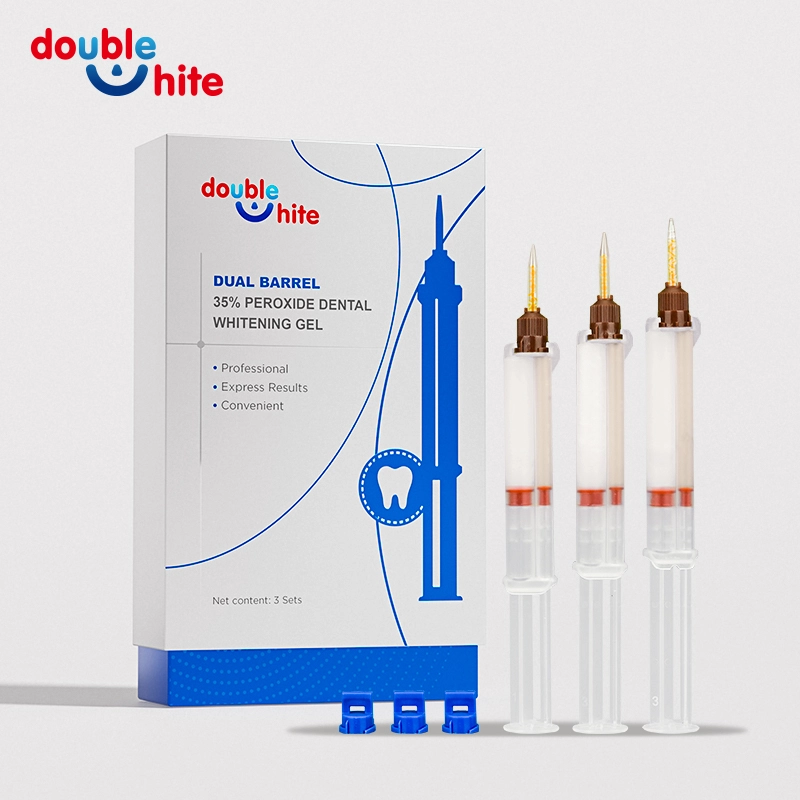
Dual barrel 35% Peroxide Dental Whitening Gel
Double White Dual Barrel 35% Peroxide Dental Whitening Gel delivers professional-grade teeth whitening with powerful, fast-acting formula. Designed for optimal results, this gel brightens your smile safely and effectively. Ideal for dental use, it ensures superior stain removal and lasting white teeth.

Hydrogen Peroxide Residue Free Teeth Whitening Strips RFHP01
Double White Hydrogen Peroxide Residue Free Teeth Whitening Strips RFHP01 deliver the best quick teeth whitening results without residue. These best teeth whitening strips offer safe, effective brightening for a confident smile. Experience one of the best teeth whitening products today.

HP Teeth Whitening Alcohol-free Strips HPNA-01
Discover Double White’s HP Teeth Whitening Alcohol-free Strips HPNA-01, the best teeth whitening strips designed for a brighter smile without irritation. Alcohol-free formula ensures gentle yet effective whitening. Achieve professional results safely and easily at home with these top-rated teeth whitening strips.

PAP Non-Alcohol teeth whitening strips OEM ODM PAPNA-03
Professional and dental grade material white strip teeth whitener helps remove stains effectively and powerfully yet gently, specially designed for sensitive teeth. FDA registered and clinically proven to effectively and safely whiten teeth.
Send us your inquiry
Reach out to us through the form below or via the contact information provided.
Our dedicated team is committed to providing prompt and personalized responses to all your queries.
Please fill out the fields above with your full name, email address, and comment.
Copyright © 2026 Double White All Rights Reserved. Designed by gooeyun













Whatsapp: +8615920313473
cndoublewhite
Doublewhite
doublewhitecn
cndoublewhite
cndoublewhite